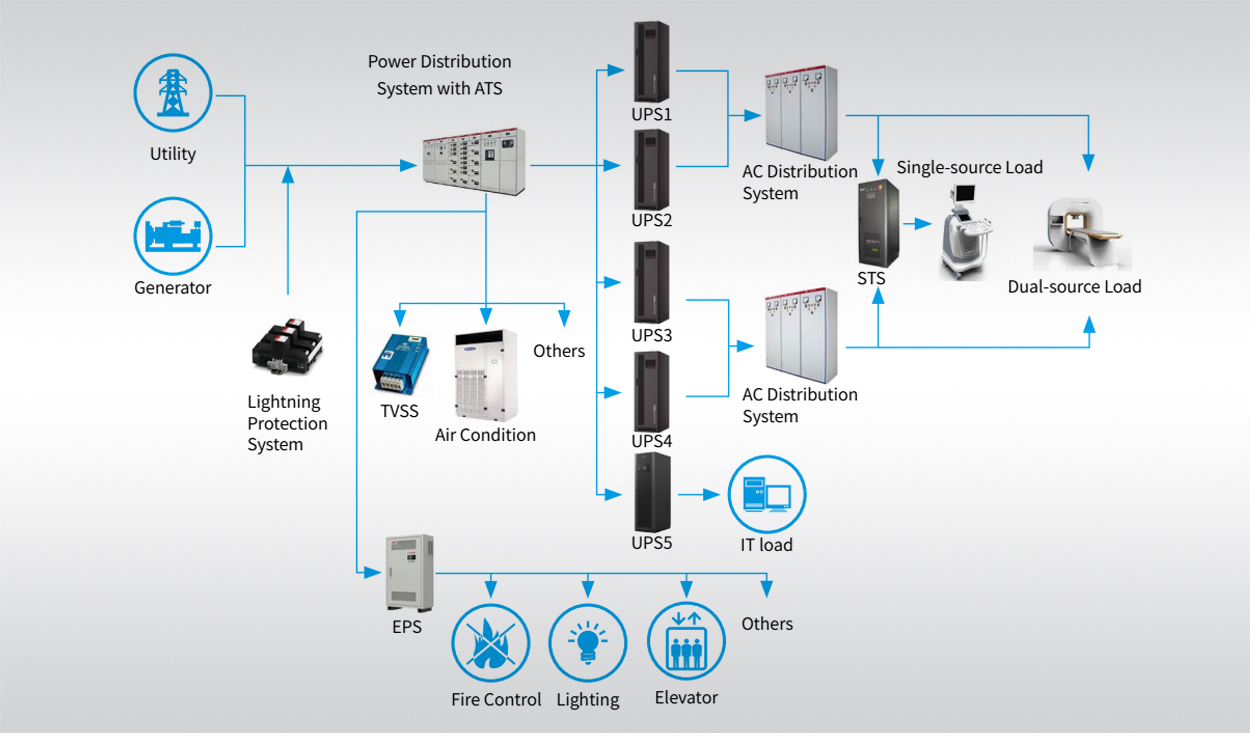
The protection of patients, reliability of equipment and extended lifecycle of components are requirements that distinguish medical devices and medically approved Power Supplies from other consumer and commercial electronics. Moreover, when the use of an uninterruptible power supply can mean the difference between success or failure in the applications of these devices, there are understandably stringent safety standards to adhere to — such as the IEC 60601-1 series of standards that apply to the performance and safety of Medical electrical equipment
In general, medical equipment must be designed with the following factors in mind:
- The device must have reinforced or doubled insulation to meet low leakage requirements.
- Components such as the AC input, Power Supply and DC output must be reliably isolated to reduce the risk of shock occurring.
- There must be further isolation around other electrical components like conductors.